Metals are located on the left of the periodic table, and nonmetals are located on the upper right. Where are the metals located on the periodic table? This brings the number of naturally occurring elements to 98. However, it turns out there are other elements that occur in trace amounts naturally. Scientists used to believe that, except for the element technetium, all the elements up to element 92 (uranium) could be found in nature. The metals consist of the alkali metals, alkaline earths, transition metals, lanthanides, and actinides.Īlso know, what percent of the elements on the periodic table are found in nature? They are grouped together in the middle to the left-hand side of the periodic table. As we move down the group from top to bottom in the group 1, the reactivity of alkali metals increases.

Alkali metals are the most reactive type of metals from the entire Periodic table of elements. Their shape can be easily changed into thin wires or sheets without breaking.įurthermore, what are all the metals on the periodic table? Most elements can be considered metals. Alkali metals are located on the left most side of the Periodic table in group 1. They are usually shiny, very dense, and only melt at high temperatures.

The periodic table on the left separates elements into three groups: the metals (green in the table), nonmetals (orange), and metalloids (blue). In this regard, are periodic tables mostly metals? Metals are located in the middle of the table, to the left of the stair-step line. The remaining elements are metalloids, which share properties of both metals and nonmetals. Predict the general properties of elements based on their location within the periodic table Identify metals, nonmetals, and metalloids by their properties and. Where are metals located on the table Click card to see definition. 24.About 80 percent of the elements are metals (shiny elements that conduct heat and electricity well), and 15 percent of the elements are nonmetals (poor conductors of heat and electricity). The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals. It also consists of non-metals, mettaloids, and. The periodic table on the left separates elements into three groups: the metals (green in the table), nonmetals (orange), and metalloids (blue). While these elements are ductile and malleable, they are not the same as. Elements in the periodic table are arranged according to their ATOMIC NUMBERS. The Table of Elements is mostly made up of metals which is located in the middle and the left side of the table. The 7 elements classified as other metals are located in groups 13, 14, and 15. Metal elements have metallic bonding where. The majority of elements in the periodic table are (METALS / nonmetals). Metals are located on the left hand side of the periodic table and comprising a large part of the periodic table. As you go down a group, the elements generally become (MORE / less) metallic.
#Where are the metals located on the periodic table series#
Elements across a series have the same number of PRINCIPAL ENERGY LEVELS. Elements within a group have a similar number of VALENCE ELECTRONS. What sublevels are filling across the Transition Elements? d AND f 19. The most active element in Group 17 is FLUORINE.
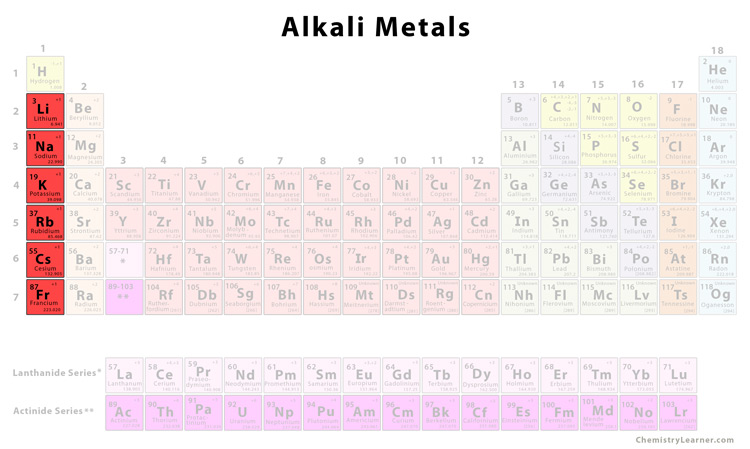
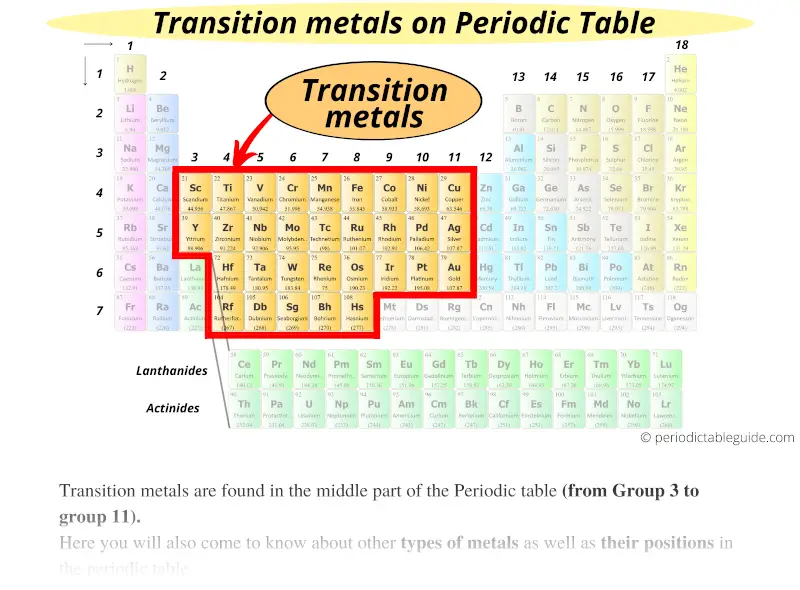
As you go from left to right across the periodic table, the elements go from (METALS / nonmetals) to (metals / NONMETALS). Group 18 elements are called NOBLE GASES. Elements of Group 3-12 are called TRANSITION METALS. On a standard periodic table, metalloids occupy a diagonal area running from boron to tellurium, in the block on the right. Elements of Group 2 are called ALKALINE EARTH METALS. If you look at the Periodic table you will find that the metal. Elements of Group 1 are called ALKALI METALS. So, because most elements of the Table are metals it makes sense to begin by looking at them. Where is the lowest electronegativity found? LOWER LEFT (Fr) 11. Where is the highest electronegativity found? UPPER RIGHT (F) 10. Why? OUTERMOST ELECTRON IS FARTHER AWAY FROM NUCLEUS SHIELDING EFFECT OF INNER ELECTRONS.
As you go down a group, the first ionization energy generally (DECREASES / increases). Why? INCREASED POSITIVE NUCLEAR CHARGE 8. As you go from left to right across a period, the first ionization energy generally (decreases / INCREASES). A positive ion is (larger / SMALLER) than its parent atom? 7. A negative ion is (LARGER / smaller) than its parent atom? 6. Why? ADDITIONAL PRINCIPAL ENERGY LEVELS 5. As you travel down a group, the atomic size (decreases / INCREASES). Why? INCREASED POSITIVE NUCLEAR CHARGE 4. As you go from left to right across a period, the atomic size (DECREASES / increases). Where are the most active non-metals located? UPPER RIGHT 3. Where are the most active metals located? LOWER LEFTĢ.
